Five tips for medtech innovators working with UHS
Medtech innovation has huge potential to improve patient care, outcomes and efficiency across the NHS. However, turning a promising idea into a technology that is tested, approved and adopted in real‑world healthcare settings can be complex and challenging.
From understanding clinical need to navigating governance and planning for adoption, success requires more than good technology alone. Here, Phil Jewell from the SETT Centre at University Hospital Southampton (UHS) shares five practical tips to help innovators move from idea to impact.
1. Start with the clinical problem, not the technology
The most successful medical technologies begin with a clearly defined clinical need. Before investing heavily in product development, ensure you fully understand the unmet need, the patient population, and the current care pathway.
Engage early with clinicians, nurses, AHPs and service managers to validate the problem and test assumptions. Most innovations which successfully reach patient care are originally developed within real-world experience and are born from questions such as: Is this issue frequent enough to justify a solution? What are the real-world constraints in practice? How will this integrate into workflow?
A solution that demonstrably improves patient outcomes, safety, experience, or efficiency, while fitting within existing NHS systems, will always have a stronger case for adoption than a technically impressive product searching for a problem.
2. Engage early with R&D and governance teams
Navigating regulatory requirements, ethics approvals, and NHS governance processes can be complex. Engaging early with your local R&D team can save significant time later in development.
At University Hospital Southampton NHS Foundation Trust, the R&D function can advise on study design, sponsorship, feasibility, and regulatory pathways. Early conversations help ensure that evaluation plans are proportionate, compliant, and aligned with NHS standards.
Building governance, as well as sponsorship, regulation and ethics considerations into your development plan from the outset avoids costly redesigns and delays. It also signals credibility to partners, investors, and potential adopters.
3. Use WHISDM to map the innovation landscape
Understanding where your technology sits, and which organisations can help you develop it within the wider system, is critical. The Wessex Healthtech Innovation Support Digital Map (WHISDM), provides a visual overview of innovation assets, expertise, facilities and support networks across the region.
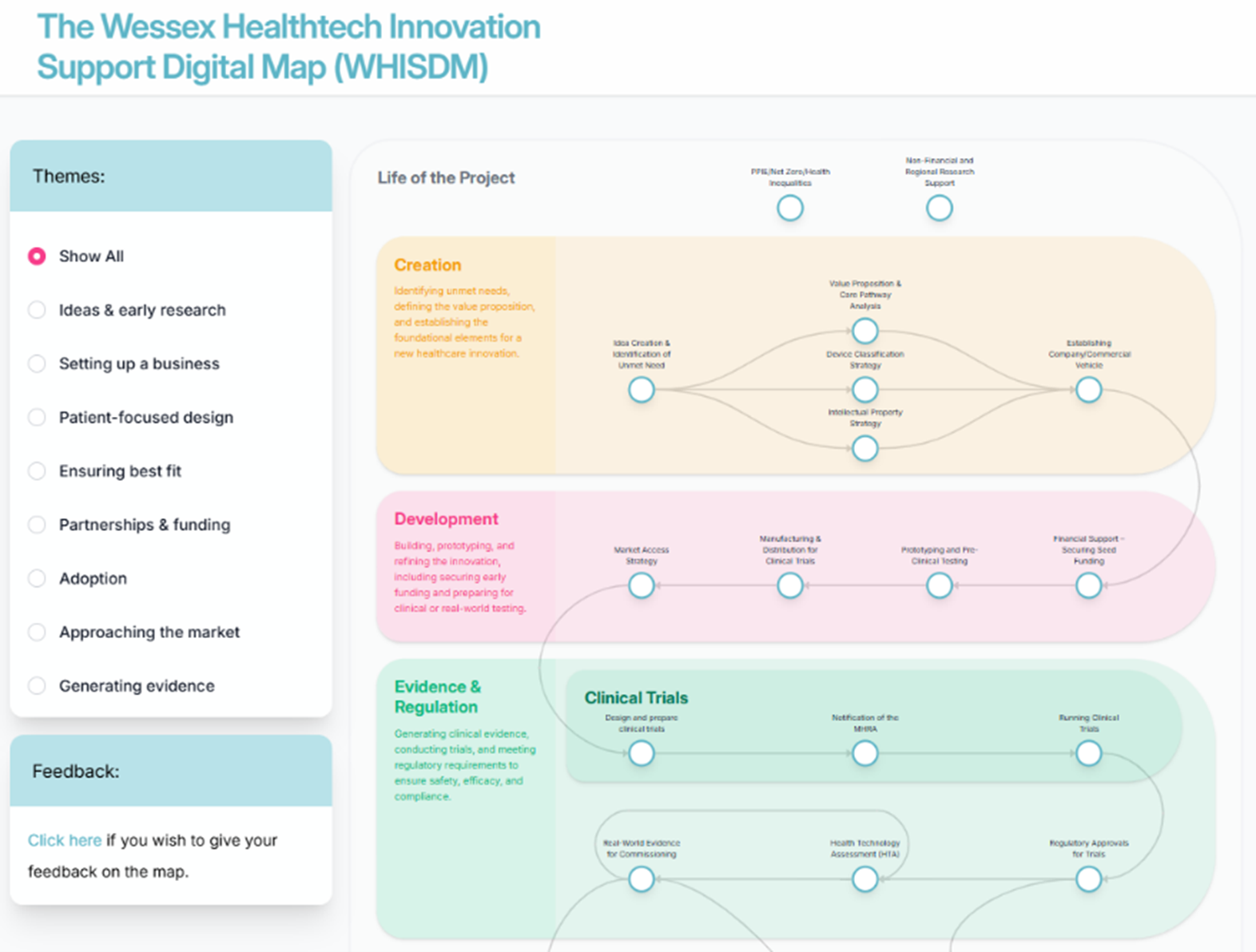
WHISDM can help you identify potential collaborators, specialist testing facilities, academic partners, and NHS contacts. For early-stage innovators in particular, it provides clarity on who does what and how to connect with them.
Rather than developing in isolation, use WHISDM to embed your project within the regional innovation ecosystem. Collaboration accelerates progress and strengthens your value proposition when seeking funding or clinical validation.
4. Build relationships through events and networking
Innovation rarely succeeds in isolation. Networking and sector events provide invaluable opportunities to test ideas, gain feedback, and form partnerships.
The recent National HealthTech Series event, hosted in Southampton by the Southampton Emerging Therapies and Technologies (SETT) Centre in conjunction with the NIHR Healthtech Research Centre in Accelerated Surgical Care, demonstrated the power of bringing together clinicians, innovators, academics and industry leaders to discuss healthcare transformation. The event received 84 attendees, the majority hailing from industry partners from across the UK, but also included representatives from other NHS trusts, universities, the NIHR, as well as local and national government.

Wessex Health Partners supported the event and their Managing Director Christine McGrath stated:
“I was delighted to welcome colleagues from across the country to the National HealthTech Series in Southampton, where the insights shared highlighted both the scale of opportunity and the critical role healthtech will play in shaping the future of UK healthcare. The day showcased national priorities, government policy, the ingenuity of innovators across our region and examples of Wessex’s academic excellence.
“Together, these conversations demonstrated the depth of talent and ambition within Wessex Health Partners and our wider Life Sciences Innovation ecosystem. My thanks to all speakers, contributors and organisers for making today such a success.”
Events such as this create space for candid conversations about adoption barriers, procurement realities and system priorities. Attending and participating in events enables you to refine your messaging, understand NHS drivers, and identify collaborators. Often, the most impactful progress happens not in formal presentations, but in the conversations that follow.
5. Plan for adoption from day one
Regulatory approval does not guarantee NHS adoption. Commissioners and providers must understand the clinical value, economic case, and operational impact of your innovation.
From early development stages, consider:
- What evidence will decision-makers require?
- Does the innovation save time, reduce cost, or improve outcomes?
- Who is the budget holder?
- How will implementation be supported?
Embedding health economics, patient feedback and insight, implementation planning, and stakeholder engagement into your development strategy significantly increases the likelihood of successful uptake. Adoption is not an end point, rather it is a process that must be actively designed.
MedTech development is a journey that benefits from collaboration, clarity, and strategic planning.
If you are developing or evaluating a new medical technology and would like to explore support pathways, the UHS SETT Centre team would be pleased to start a conversation. Get in touch with us at sett@uhs.nhs.uk.